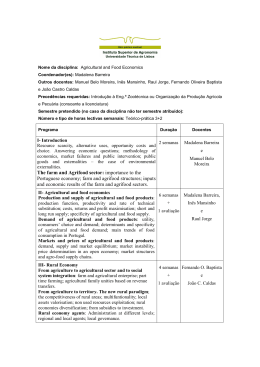
training programme in pharmaceutical medicine safety and risk management INFARMED, Lisbon 08 - 10 march 2012 safety and risk management 08 – 10 MARch 2012 LocaL: INFARMED, Lisbon curricuLar unit Leader: teresa Herdeiro, Pharmd, MPhil, Phd Lecturers: Ana Araújo, Madalena Arriegas, Salvatore Curti, Germano Ferreira, Helena Gama, Teresa Herdeiro, Sandra Madaleno, Alexandra Pêgo, Dirk Teuwen, Inês Vaz. organisation: Masters in Pharmaceutical Medicine Post-Graduate course in Pharmaceutical Medicine Director: Luis Almeida, MD, PhD, FFPM ([email protected]) Deputy Director: Bruno Gago, PharmD, PhD (bmgago@ ua.pt) address: Health Sciences Department University of Aveiro Campus Universitário de Santiago | Edifício III 3810-193 Aveiro | Portugal Tel. +351 234 370 213 | Fax +351 234 401 597 | E-mail: [email protected] | http://www.cs.ua.pt DAy 01 08 MARCH time Programme Lecturer 10h00-10h30 0. introduction Teresa Herdeiro (University of Aveiro) 1. History and background of Pharmacovigilance 10h30-12h00 1.1. History; 1.2. The WHO and safety reporting; 1.3. ICH and CIOMS initiatives. CIOMS – function and purpose; 1.4. Legal basis in the EU and the US; 1.5. New European Pharmacovigilance legislaton Ana Araújo (INFARMED) 12h00-13h00 1.6. Assessment and classification of adverse events (AEs), adverse drug reactions (ADRs). Serious Adverse Events (SAEs) and Suspected Unexpected Serious Adverse Reactions (SUSARs); evidence for association and causality; 1.7. The concept of benefit / risk assessment. determination of causal relationship between the medicinal product and the adverse event. Inês Vaz (UFN - Unidade de Farmacovigilância do Norte) Lunch 13h00-14h30 2. 14h30-16h30 16h30-17h00 17h00-19h00 collection, management and reporting of safety information for investigational products 2.1. Drug safety during clinical trials; 2.2 Collection of AEs in clinical trials; 2.3. Role of sponsors and investigators in reporting: regulatory requirements Dirk Teuwen & Salvatore Curti (UCB) Coffee-break 2.4. Predisposing factors in health and disease; 2.5. Using MedDRA terms; 2.6. EU and US regulations concerning safety data reporting and data management. Dirk Teuwen & Salvatore Curti (UCB) DAy 02 09 MARCH time Programme 3. Lecturer collection and management of safety information for marketed products 09h00-11h00 3.1. Why do we need Risk Management Plans (RMP)? 3.2. Legal Basis in EU and the US 3.3. Requirements for a RMP 3.4. The EU-RMP template 11h00-11h30 Coffee-break 11h30-13h00 3.5. Pharmacovigilance Studies, designing, conducting and reporting Madalena Arriegas (European post-authorization studies Medicines Agency) 3.6.Risk Minimization Activities 3.7. Assessment and monitoring compliance 3.8. Case Studies 13h00-14h30 Lunch 4. 14h30-16h00 Madalena Arriegas (European Medicines Agency) Safety information to regulatory authorities 4.1. Sources of safety information 4.2. Post-marketing safety requirements and spontaneous expedited reporting in the EU and the US. 4.3. Electronic transmission Ana Araújo (INFARMED) 4.4. Dosage, accumulation, medication errors and interactions; drug adherence/compliance. Sandra Madaleno (Pfizer) 16h00-16h30 Coffee-break 16h30-18h00 4.5. Periodic reporting in the EU and the US 4.6. Safety evaluation and implementation of safety measures: Revocation, suspension, USR, SPC variation, communication to HCP and general public. Alexandra Pêgo (INFARMED) DAy 03 10 MARCH time Programme 5. company’s drug safety and pharmacovigilance system 09h00-11h00 5.1. Company responsibilities for pharmacovigilance. 5.2. Pharmacovigilance and contractual agreements with third parties. 5.3. The Qualified Person for Pharmacovigilance (QPPV) in the European Economic Area. 5.4. Designing the company’s processes: SOPs. 11h00-11h30 Coffee-break 11h30-13h00 5.5. Pharmacovigilance inspections. 5.6. Crisis management. 5.7. Risk communication. 13h00-14h30 Lunch 6. Lecturer Helena Gama (Bial) Helena Gama (Bial) evaluating safety information and developing product labelling 14h00-17h00 6.1. Definition of a safety signal. 6.2. Regulatory requirements for signal detection, interpretation and management. 6.3. Sources of signals. 6.4. Qualitative and quantitative methods of signal detection. 6.5 Signal evaluation, developing and managing company core safety information and product labeling Germano Ferreira (Sanofi-Pasteur) 17h00-17h30 The role of the pharmaceutical professional in drug safety and pharmacovigilance. Alexandra Pêgo (INFAMED), Helena Gama (Bial), Teresa Herdeiro (UA) Module objectives: • To provide an overview of medicinal products safety, from development to marketing. • To present the principles and practice of pharmacovigilance, and the roles of the various authorities/entities involved. • To know post-marketing safety requirements and spontaneous reporting/electronic transmission/periodic reporting. • To evaluate the sources of signals and to know the regulatory requirements for signal detection, interpretation and management. Learning outcomes of the Module: By the end of this module participants will be able to: • Understand the need of assessing medicinal products safety, both during development and the marketing period. • Identify and classify adverse events and adverse drug reactions, evaluating the causality. • Develop Risk Management Plans and Risk Minimization Activities, while considering their legal and regulatory frameworks. • Understand the procedures for Expedited Reporting Notification, both in the EU and in the US, and periodic safety reporting. • Assess drug safety information and identify options for action in response to safety signals. • Discuss issues surrounding effective crisis management and the communication of drug safety issues to Regulatory Authorities, Healthcare Professionals and the General Public. • Develop pharmacoepidemiological studies and methods to evaluate drug safety. Endorsed by the Associação de Médicos Portugueses da Indústria Farmacêutica
Baixar